Imagine if matter could go through other matter. Wouldn’t the world be a different place?
Let’s say you went on a hiking trip and arrived at a bridge over a river. Theoretically, you have two options to get to the other side: you could either go over the bridge, or you could paddle under it. But what if you could go through the bridge?
This is analogous to a phenomenon in quantum physics — physics at very small length scales — known as quantum tunnelling. A recent Nature publication highlights a discovery made on quantum tunnelling by Dr. Aephraim Steinberg and his team at the University of Toronto’s Department of Physics.
The team proved that quantum tunnelling is not instantaneous. In other words, if a particle were to go through a barrier, it would spend a certain amount of time in the barrier. According to Steinberg, the most surprising thing about the principle of quantum tunnelling is that a particle going through a barrier need not be an electron.
“In principle, a baseball could do this,” he said in an interview with The Varsity.
What is quantum tunnelling?
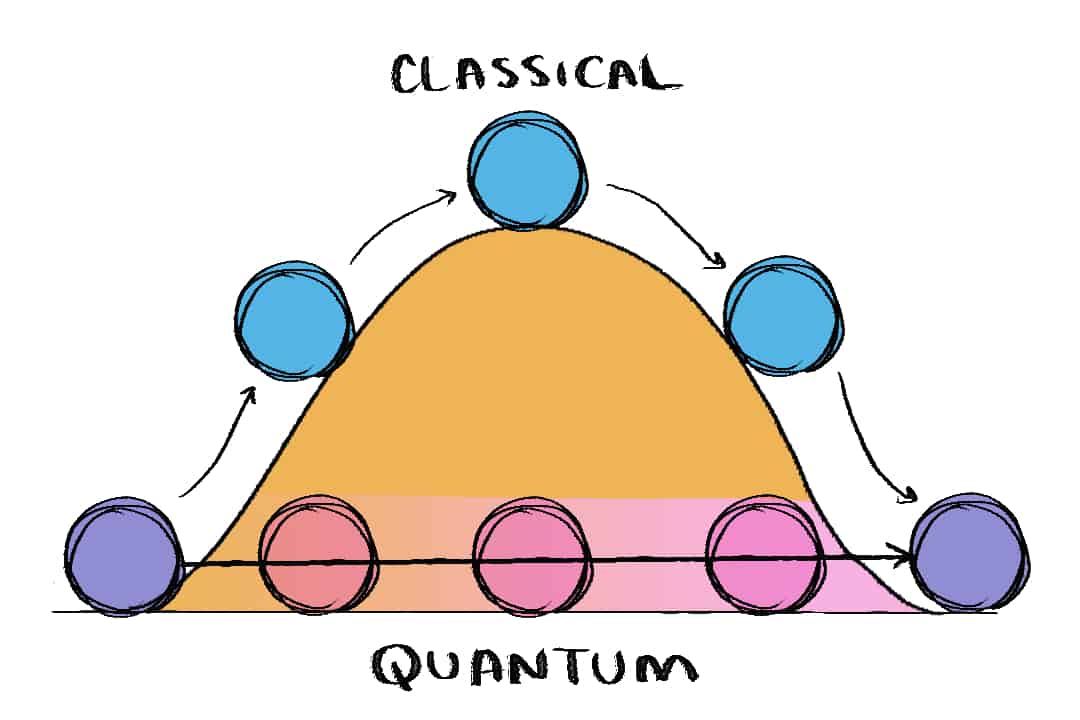
Most physicists explain quantum tunnelling with a classic analogy of a ball and a hill. If you try to push a ball up a hill and you give it a strong push, it won’t get over the crest of the hill unless you give it one big push so that it can reach the hilltop. In physics talk, you need to make sure that the ball has enough kinetic energy to overcome the hill’s potential energy.
However, the quantum world doesn’t use those seemingly logical rules — and it is for this reason that the field of quantum mechanics can be so difficult to explain.
Unlike classical physics, quantum physics is probabilistic. That means that instead of calculating the exact locations and positions of particles, quantum physicists investigate the probability of electrons being located in a certain area.
If you had a quantum ball and hill scenario, even though it is highly likely that you’ll find the ball on your side of the hill, there’s still a tiny probability that you’ll find your ball inside the hill. There’s an even smaller chance that your ball could eventually end up on the other side of the hill — almost as if it had dug a tunnel through.
This is how quantum tunnelling is observed in most experiments: a change in location from one side of a barrier to another.
According to Steinberg, he wouldn’t call his recent findings ‘a discovery.’
“I think very few people thought the process was instantaneous,” he said. “There’s been over 80 years of debate over how exactly one should talk about the duration and process of different attempts to define and to measure it. And… a year or two ago, there was a new story with another group, and they found that it was instantaneous. And we disagreed with that description.”
How did Steinberg’s team do it?
The team measured how long a rubidium atom spends inside a barrier of light constructed using lasers and microwaves. Rubidium atoms were cooled to one nanokelvin and directed by lasers to move in one direction. The path of these atoms was then blocked by another laser beam, which served as a barrier of light. The experimenters wanted to know how long the atoms spent inside the barrier before making it out on the other side.
So, how do you measure the travel time of an atom?
“If you let every particle carry its own stopwatch, and then you wait and look through to see when the particle appears on the other side, you can look at its stopwatch [to measure the time it took],” Steinberg said. “It turns out that particles like electrons and atoms have a property called spin… and the spins… react to magnetic fields.”
Steinberg and his colleagues used the spins of the rubidium atoms as hands on a clock face. Inside the light barrier, they exposed the atoms to a magnetic field, which caused the ‘clock hands’ to rotate — effectively revealing how long each atom spent inside. The longer the particle took inside the barrier, the longer it was exposed to the magnetic field, and the more its spin rotated.
Ultimately, the researchers found that each atom spends 0.61 milliseconds inside the light barrier — a small number, but definitely not zero.
“Basically, what we’re really striving for is simply a better understanding of how the quantum mechanical particles get where they get,” Steinberg said. “Tunnelling is one of the most striking examples of this. Honestly, what interests me is the modern question of how we think about this quantum world at all.”
Quantum tunnelling is a process we see in our everyday lives.
“[It has a role in] things like photosynthesis, even vision, where electrons and even protons are required in order to transfer energy along some of the biochemical pathways,” Steinberg said. “And maybe one of the most fundamental [roles quantum tunnelling has] in our existence is that a lot of nuclear processes require tunnelling. So the fusion that occurs in the sun that allows us to survive actually relies on tunnelling as well.”
What’s next for the team?
Steinberg noted that his team is going in “a few different directions.”
“It’s relatively straightforward to just calculate — if I throw a lot of particles in a barrier — on average, how much time [they would spend] in the barrier. That’s not so difficult,” Steinberg said. “What makes the process confusing comes back to this indeterminism in quantum mechanics; some particles get through and others are reflected. People wanted to know, ‘How long does it take just for the particles to get through?’ ”
But the team was in for a surprise. In the particular place they looked, transmitted and reflected particles spent the same amount of time in the barrier. Steinberg is now interested in asking different questions.
“Instead of just asking, ‘How much time do the particles spend overall in the barrier?’ you ask more specific questions. Where in the barrier are they spending most of their time? Are they on the left hand side or on the right hand side or middle?” Steinberg said. “Then, we expect to actually see different results with the transmitted and the reflected particles. So we want to refine our experiments to do that.”
The probabilistic and often controversial nature of quantum physics has given the physicist a lot to think about.
“Although the predictions for what we’re used to asking are probabilistic, the laws that we use for our predictions are, in fact, completely deterministic,” Steinberg said. “In other words, we can predict exactly what the final probability distribution is — there’s no ambiguity about that.”
Steinberg continued, “So it’s a controversy that’s more on the philosophical side. What does it mean to have a description of nature that only gives us probabilities? And does it give us definite results?”