Microbes are everywhere. Profuse and diverse, they are tightly connected to our lives. Some of them are unfriendly, such as the virus that curses us with the common cold. Others perform vital functions for us, such as the gut bacteria that help break down proteins, lipids, and carbohydrates in our diets.
Despite the importance of microbes in our world, we know remarkably little about their evolution. Microbes don’t evolve in isolation from other organisms; in fact, they often collaborate and form mutually beneficial relationships — or symbioses — with host creatures.
Many fundamental questions about microbes remain unanswered, including whether the symbioses emerge because hosts ‘choose’ the most beneficial microbes or because the microbes adapt to benefit their hosts. And how do these microbial symbioses evolve on a genetic level?
Rebecca Batstone, a postdoctoral fellow at the University of Illinois, addressed some of these questions during her time at U of T as a PhD student in the Frederickson lab in the Department of Ecology & Evolutionary Biology. As part of her research, she looked to legumes and underground symbiotic bacteria for answers. Her findings were recognized in the journal Science last month.
Bean science
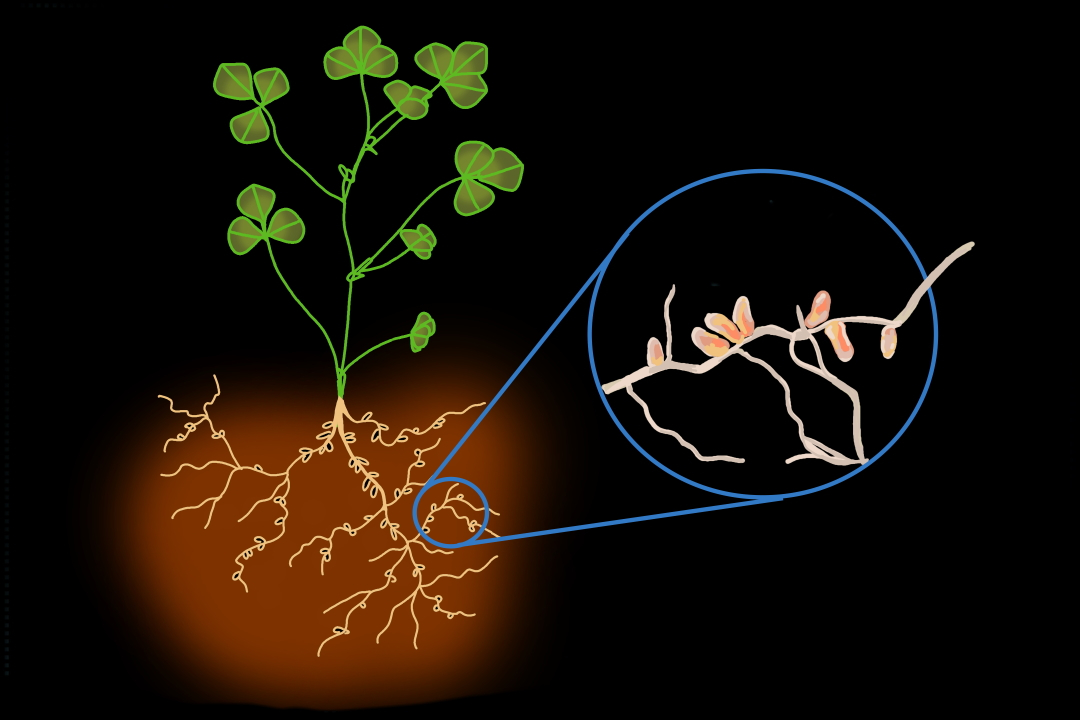
Legumes — a family of plants that includes alfalfa, clover, beans, peas, and peanuts — are extremely nutritious and protein-rich foods for a reason. Beneath our feet, amongst the tangles of legume roots in the soil, tiny nitrogen-fixing bacteria are doing something plants can’t do on their own: converting atmospheric nitrogen into biologically usable ammonia. These bacteria — known as rhizobia — give legumes a nitrogen boost in exchange for a home and a steady stream of sugar.
In this symbiosis, rhizobia house themselves on legume roots in little compartments called nodules. If you are ever doing some weeding in your garden or decide to dig up a patch of red clover on a U of T lawn, you’ll be able to see these for yourself. Nodules look like tiny lumps growing on the roots and are visible to the naked eye.
It is not clear what the more important evolutionary force in shaping microbe-host symbioses is. It could be the host ‘choosing’ the most cooperative partners by rewarding them with the most sugar, or microbes evolving to be better partners on their own.
To unravel the mystery, Batstone and her colleagues undertook a greenhouse experiment involving the rhizobium Ensifer meliloti and the Mediterranean legume Medicago truncatula — a popular model organism used in biology.
They added two strains of nitrogen-fixing bacteria — an effective, high-performing strain and an ineffective, low-performing strain — to five different plant varieties. Each plant variety differed in how strongly it chose bacterial partners.
Adaptive microbes, not choosy hosts
If a plant’s choosiness is the main evolutionary force shaping microbial cooperation, then we’d expect the choosiest plant variety to most strongly favour the better partner. But the ineffective nitrogen-fixer went nearly extinct across all plants, even on plant varieties that don’t discriminate much between effective and ineffective partners.
Microbes seemed to be the organism shaping the cooperation, not the plants. Independent of host choosiness, the better rhizobial partner still adapted to its local plant variety. Batstone and her colleagues made sure to sequence the rhizobial genomes before and after the evolution experiment to track the kinds of genetic changes that took place. The scientists confirmed that these mutations actually increased the strength of the mutualism in a follow-up experiment.
“When we put microbes from the beginning and the end of the experiment back onto hosts, we found they did best with the same hosts they evolved on, suggesting they adapted to their local host,” Batstone said in an interview with phys.org. “The derived microbes were more beneficial when they shared an evolutionary history with their host.”
Therefore, cooperation depended on the match between partners, and cooperation increased as bacteria adapted to their hosts. This result is exciting because it suggests that there might be ways to create microbes that better benefit their hosts.
“It’d be kind of cool, down the road, if we could figure out ways to engineer our microbiomes to select on traits that we want,” said Batstone in an interview with The Varsity. “But from our research, what it’s really suggesting is that there’s probably no universally great microbe, or even microbiome… It really does depend on the genotype of the individual.”